Biotech Digital Brief
Report Brief
THE POTENTIAL IMPACT OF MEDICAL BIOTECHNOLOGY ON THE FUTURE OF ADULT LEARNING
FINDINGS FROM ACTION RESEARCH
Action research report carried out by:
Learning could soon become more personalized and responsive to individual biological profiles.
At the International Training Centre of the ILO (ITCILO), we are exploring the potential impact of medical biotechnology on the future of adult learning, helping it becomes more flexible, inclusive, and focused on people’s needs.
ITCILO recognizes biotechnology as a strategic domain shaping the future of work, health systems, and national sovereignty. As part of its mission to anticipate emerging skills and promote social justice, this Action Research examines how medical biotechnology —strengthened by AI, data infrastructures, and personalized health technologies - will redefine adult-learning ecosystems through 2030.
1. Introduction
Biotechnology operates at the intersection of Biology, Chemistry, Physics and Engineering, where bioprocesses translate molecular knowledge into tangible applications.
Biotechnology must be understood as a convergent technological domain whose capabilities and challenges are tightly linked to the maturity and interoperability of its enabling layers. One of the most fundamental research strands of biotechnology is to lay the groundwork for decoding, manipulating, and redesigning biological processes at multiple scales, from genes and enzymes to entire ecosystems.
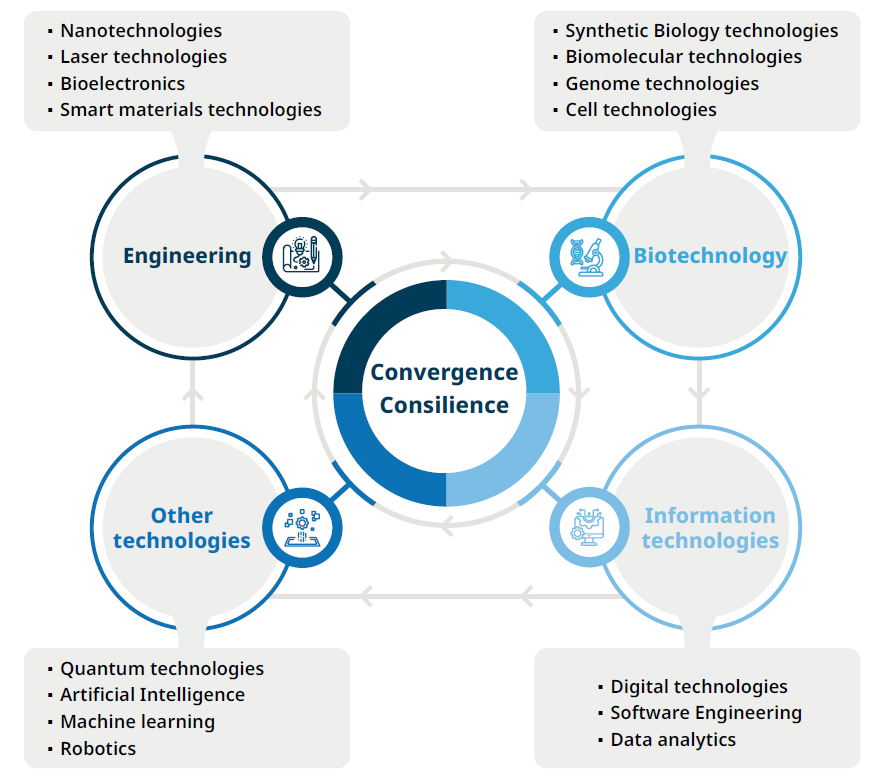
Bioinnovation. Advanced biological, physical, information, and engineering technologies integrate to form the foundation of bioinnovation, illustrating how convergent disciplines collectively drive new solutions, applications, and technological progress. (Source: adapted from Pretorius et al. 2025).
Over the past two decades, the biotechnology sector has evolved from a niche scientific endeavor into a multi-trillion-dollar driver of the global economy, spanning pharmaceuticals, agriculture, and industrial processing.
The accelerated translation of fundamental research into applied biotechnology has been fueled by advances in genomics, AI-driven drug discoveries, and high-throughput biomanufacturing. Importantly, private and governmental investments have played a key role in consolidating biotechnology as a pillar of national innovation agendas.
ITCILO recognizes biotechnology as a strategic domain shaping the future of work, health systems, and national sovereignty. As part of its mission to anticipate emerging skills and promote social justice, this Action Research examines how medical biotechnology – strengthened by AI, data infrastructures, and personalized health technologies - will redefine adult-learning ecosystems through 2030.
The present workstream focuses on refining ITCILO’s taxonomy of emerging technologies as a practical tool for guiding research activities, specifically with respect to biotechnology and its expanding impact across multiple sectors. Strengthening biotechnology literacy, skills readiness, and workforce resilience are central themes of this report.
2. Methodology
The methodology for this report integrates analysis of secondary data sources with insights gathered from key informant interviews. Secondary sources include national and regional biotechnology strategies, corporate roadmaps from leading biotechnology, pharmaceutical, and life-science companies, multiple international technology taxonomies, markets and foresight reports and additional online resources, academic publications, policy briefs and industry analyses cited throughout the document.
PART I. SITUATING BIOTECHNOLOGY IN THE TAXONOMY OF FOUNDATIONAL TECHNOLOGIES
Part I of this research provides an integrated analysis and mapping of the enabling innovations that underpin the contemporary expansion of the biotechnology sector and its diverse products.
PART II. DEMOCRATIZATION OF BIOTECHNOLOGY: EMERGING BIOTECHNOLOGY DOMAINS WITH HIGH IMPACT ON LEARNI
In Part II, we identify the major biotechnology strands with the greatest potential impact on adult learning, building directly on the analysis developed in Part I. This chapter distinguishes between clinical and nonclinical domains of medical biotechnology, highlighting their respective trajectories, use cases, and implications for skills development and adult learning. This framing enables clearer anticipation of the new demands that AI will place on workforces and education systems-an issue further explored in Part III.
PART III. IN-DEPTH ANALYSIS OF PRIORITIZED BIOTECHNOLOGY TRENDS WITH POTENTIAL IMPACT ON ADULT LEARN
In Part III, we will provide a deeper assessment of these priority biotechnology trends through the lens of ITCILO’s mission, considering their learning potential, ethical and governance implications and relevance for future skills development. The aim is to identify actionable opportunities that ITCILO may pursue toward 2030 and beyond, equipping workers, institutions, and policymakers with the competencies required to navigate a world in which biotechnology is increasingly embedded across healthcare systems, markets, and society at large.
To fully realize the benefits of biotechnology in education, while avoiding the amplification of risks, this action research emphasizes the need for evidence-based foundations, rigorous biosafety and ethical norms, and strong pedagogical frameworks.
3. Findings
Biotechnology has transitioned from fundamental biological research to a strategic industrial sector with profound implications for health, food systems and the environment. Supported by large-scale R&D funding, rapid innovation in genomics and bioprocessing and expanding consumer markets, the biotechnology industry exemplifies how science-driven innovation can deliver scalable solutions to 21st-century challenges.
3.1 Summary of Modifications to the ITCILO Biotechnology Taxonomy
We revised the original ITCILO taxonomy to make it more accurate, future-oriented, and aligned to learning and workforce development needs. The updated version distinguishes between clinical and non-clinical medical biotechnology and introduces additional domains that are fundamental to today’s biotechnology ecosystem, including genome sequencing, gene editing and gene therapies, structural biology, protein engineering and synthetic biology.
In the newly proposed taxonomy, it becomes clearer how biotechnology innovations are increasingly woven into the everyday technological environment, shaping consumer products, workplace practices, and even general digital literacy. Biotechnology is no longer confined to specialized laboratories: it is progressively embedded in common tools, services and processes encountered in daily life.
Biotechnology Updated Taxonomy
- Bioinformatics
- Genomics and personalized medicine
- Gene editing and gene therapy
- RNA vaccines
- Advanced Diagnostics
- Immunotherapy
- Genetic testing
- Microbiome
- Stem cells
- Structural Biology
- Synthetic Biology
- Protein Engineering
- Agricultural Biotechnology applications
- Environmental Biotechnology applications
- Industrial Biotechnology applications
3.2 Bioinformatics: Data Standardization, Accessibility, AI Integration, and Adult Learning Needs.
Over the next five to ten years, the transformative potential of bioinformatics will depend not only on the availability of large biological datasets but on the ability to standardize their annotation and perform integrative analysis across multiple domains. Standardization is becoming a strategic priority: without harmonized metadata, consistent ontologies, and interoperable formats, even the most advanced AI systems cannot extract reliable insights. As biological data proliferates, AI-driven tools will be essential for unifying these datasets, identifying hidden patterns, generating predictive models, and supporting decision-making in research, healthcare, and bio-manufacturing.
This evolution has direct implications for adult learning. The future biotechnology workforce will require not only basic data literacy but also the capacity to work with standardized datasets, understand AI-assisted analytical pipelines, and critically interpret integrative multi-omics outputs. Adult learning systems must therefore adopt flexible, modular, and practice-oriented training approaches that expose learners to real datasets, annotation standards, ML workflows, and ethical considerations surrounding data use. Building these competencies will be vital for ensuring an inclusive, capable workforce for the next decade of biotechnology innovation.
3.3 Clinical Medical Biotechnology as the Core Engine of Biotechnological Innovation
Medical biotechnology remains the principal engine of biotechnological innovation, driving advances that shape research priorities, investment flows, and global health strategies. These developments often diffuse beyond medicine into agriculture, industry, and environmental applications, reinforcing the medical domain as the primary source of transformative solutions across the bioeconomy.
3.3.1 AI- Enabled Precision Medicine and Emerging Competences
Over the last decade, new techniques, particularly advanced AI methods, have reshaped clinical medical practice. These innovations accelerate drug development, enable interventions against previously “undruggable” molecular targets, revolutionize diagnostics, and open therapeutic pathways for genetic and metabolic diseases that were once considered untreatable.
The direct impact of these techniques on learning is substantial. As integrative AI analyses become embedded in diagnostics and new clinical techniques emerge, the information that individuals receive about their health will grow significantly more complex. As data collection and computational methods improve, this synergy could enable earlier disease detection, better risk stratification, more precise and effective therapies, and optimized preventative care, thereby increase treatment effectiveness and potentially reducing healthcare costs at a population scale.
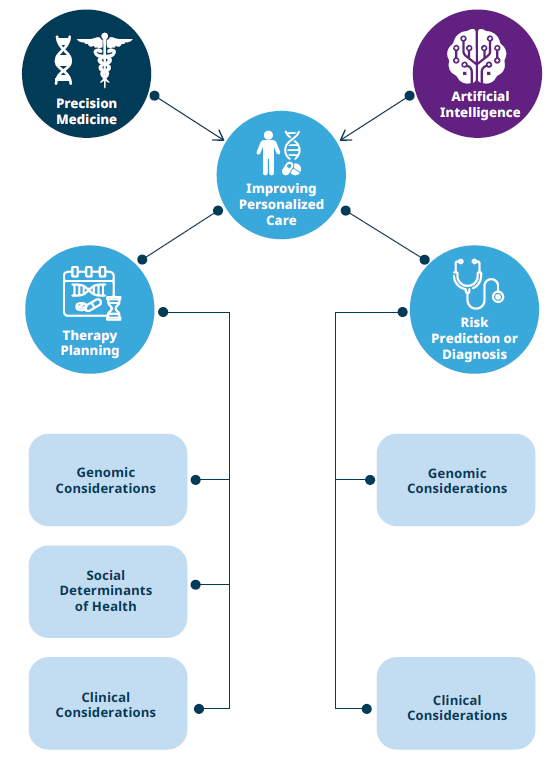
Precision medicine and AI intersect across major dimensions that advance personalized care. (Source: Johnson et al., 2021).
3.3.2 Direct use of AI and ML in drug discovery pipelines
Despite their immense promise, critical challenges remain. AI models depend heavily on the quality, diversity, and completeness of the datasets used for training; biases or gaps in underlying biological and chemical data can reduce predictive accuracy or lead to erroneous conclusions. Furthermore, ensuring the interpretability and transparency of AI-driven decisions is vital-both for regulatory acceptance and for building trust among scientists and clinicians.
Ethical considerations, including data privacy and responsible deployment of AI-generated insights, are increasingly important as computational tools move closer to influencing clinical decision-making. Over the coming decade, advances such as improved data standardization, secure and interoperable computation, and the growth of interdisciplinary expertise will be essential to fully integrate AI into drug discovery pipelines.
3.3.3 Modern Genomics and Its expanding role in Research, Medicine, and Public Health
Genomics has undergone a dramatic technological transformation over the last two decades, propelled by breakthroughs that have reshaped both research and clinical practice. Genome sequencing is rapidly becoming more affordable and accessible, accelerating its integration into research, public health, and routine clinical care.
Despite enormous progress, genomics continues to face significant scientific, ethical, and societal challenges that shape its future impact. Interpreting genomic variation remains another major bottleneck, as a small fraction of detected variants has clear clinical significance; many remain classified as variants of uncertain significance, and limited understanding of gene–gene interactions and phenotype effects further constrains interpretation. Equity and representation pose additional concerns, as most genomic databases disproportionately include individuals of European ancestry, introducing bias into predictive models and reducing diagnostic accuracy for underrepresented populations.
3.3.4 Gene Therapy and Gene Editing: Transforming Medicine at the Molecular level
Looking forward, gene therapy and gene editing are rapidly evolving toward more sophisticated, programmable, and safer interventions. Next-generation tools such as base and prime editors offer the possibility of correcting mutations with minimal collateral damage, while epigenetic editing introduces reversible ways to modulate gene expression without altering the underlying DNA sequence. The field is also moving toward integrated therapeutic strategies, combining gene editing with cell therapies, RNA therapeutics, or synthetic biology approaches to create living medicines capable of sensing and responding to disease signals.
Gene therapy and genome editing thus stand at the threshold of a new era-one in which medicine transitions from treating disease to actively engineering health, driven by unprecedented precision in how we can modify, program, and sustain biological systems.
3.3.5 RNA-Based Therapeutics
Current challenges in the RNA therapeutics field center on delivery efficiency, molecular stability, and immunogenicity. Achieving targeted delivery remains one of the major bottlenecks: while lipid nanoparticles (LNPs) have enabled first-generation mRNA vaccines, next-generation delivery systems will require enhanced tissue specificity, reduced off-target effects, and improved safety profiles.
From a policy and governance perspective, professionals must be prepared to assess challenges associated with equitable access, genetic data stewardship, global supply chains, and pandemic readiness manufacturing, issues that the COVID-19 crisis brought sharply into focus. Together, these technical and regulatory demands show that RNA-based medicines are not only a rapidly advancing scientific frontier but also a powerful driver for rethinking training systems, cross disciplinary competencies, and adult-learning strategies across the biotechnology sector.
3.3.6 Advanced Diagnostics
Diagnostic technologies are evolving rapidly, driven by advances in molecular biology, point of-care platforms, and AI-enabled analytical tools. The paper highlights that, despite these scientific and technological gains, the regulatory landscape for diagnostics remains highly uneven across countries, resulting in fragmented standards, variable product quality, and uneven global access. Unlike pharmaceuticals, which benefit from well-established regulatory pathways, diagnostics encompass a wide and heterogeneous range of risk profiles, from simple lateral-flow tests to complex digital-AI systems, requiring regulatory frameworks that are agile, proportionate, and context-specific.
3.4 Non-clinical Medical Biotechnology and the Rise of Accessible Bio-Products
At the professional level, clinicians, nutritionists, health coaches, and regulatory professionals will require new competencies to evaluate microbiome reports, counsel consumers, and navigate rapidly changing standards for data accuracy, privacy, and ethical use. More broadly, consumer-facing microbiome technologies highlight a wider societal shift toward personalized, data-centric health management, underscoring the need for continuous learning systems that equip adults with the knowledge to critically interpret biological data, make informed health decisions, and participate meaningfully in emerging models of preventive and participatory healthcare.
3.5 Agricultural Biotechnology
Biotechnology companies are increasingly integrating AI and ML tools into autonomous robotic platforms capable of performing essential agricultural activities, such as crop harvesting, at significantly higher speed and efficiency than manual labor. Computer vision systems combined with DL algorithms are used to process and analyze high-resolution imagery collected by drones, enabling real-time monitoring of crop status, soil conditions, and potential stress factors. Furthermore, ML-based predictive models support decision-making by tracking and forecasting environmental variables - including weather patterns - that directly influence crop performance and yield outcomes.
3.6 Food Biotechnology
Technological advances have largely taken place in areas such as genetics, metabolic engineering, precision fermentation, and enzyme- or cell-based bioprocesses, ultimately aiming to improve food ingredients and final products, as well as to enable novel and more sustainable alternatives.
AI has become an important tool in the food sector, supporting activities such as process modeling, quality prediction and control, sensory evaluation, drying optimization, and addressing complex food processing challenges.
AI also holds significant promise for biotechnology-driven innovation in the food sector. For example, enzyme development can be accelerated through AI by enabling the design and modeling of complex biochemical reactions, thereby facilitating the creation of novel or improved food ingredients and processing solutions.
3.7 Industrial Biotechnology
The faster development of new biotechnological processes for chemical production has been driven by major advances in industrial biotechnology, metabolic engineering, and synthetic biology.
The rapid and precise engineering and characterization of microbial cell factories, together with advances in data acquisition, analysis, and integration, are positioning biotechnology closer to traditional engineering disciplines with increasingly rational and predictable design.
3.8 Marine Biotechnology
Despite its vast potential, the marine environment remains largely unexplored, with more than 90% of its biodiversity still unknown. Scientific progress is limited by the difficulty of accessing deep-sea habitats, challenges in taxonomic classification, and the inability to reproduce isolation and characterization of bioactive compounds. Emerging technologies, including remotely operated vehicles, automated data collection, advanced analytical methods, and modern high-throughput screening, are helping overcome these barriers by enabling efficient sampling, rapid compound testing, and more targeted drug discovery.
3.9 Environmental Biotechnology
As environmental challenges grow in scale and complexity, AI is emerging as a transformative tool to strengthen these biotechnological strategies through predictive modeling of pollutant degradation, optimization of bioprocess parameters, modeling of microbial community dynamics, and real-time environmental monitoring and control.
Recent advances in AI have greatly accelerated microbial bioremediation by enabling the rapid discovery of pollutant-degrading microorganisms and the design of more efficient enzymes. AI has also expanded capabilities for detecting and monitoring environmental contaminants through non-invasive, real-time analytical approaches. Additionally, unclear regulatory standards for using AI-guided microbial systems in natural environments create.
4. Implications
In biotech, techniques are transversal and generate impact across multiple domains simultaneously. For ITCILO, recognizing this dynamic and multidimensional landscape will be essential for shaping future-ready adult learning strategies, ensuring professionals are prepared not only to use individual technologies but to navigate the growing intersections between them.
4.1 Restoring Cognition: Gene and Cell-Based Innovations for Neurodevelopment and Neurodegeneration
Continued advances in gene therapy, precision medicine, and innovative delivery modalities are expected to improve quality of life, preserve cognitive and motor function, and reduce long-term disability for affected individuals. Earlier and more accurate diagnoses, supported by genomics, advanced imaging, and AI-enabled decision tools, will be essential to ensure timely access to emerging therapies and maximize therapeutic benefit. Collectively, these developments suggest a future in which debilitating central nervous system (CNS) diseases can be managed more effectively, enabling greater participation in education, employment, and community life throughout the lifespan.
This progress also creates new opportunities for ITCILO and other capacity-building institutions to design tools, training programs and initiative-taking methodologies that anticipate evolving skills needs in healthcare systems and the wider labor market. By systematically monitoring breakthroughs in biotechnology and integrating them into adult-learning strategies, ITCILO can help ensure that professionals, patients, and caregivers are well prepared to understand and benefit from innovation. Furthermore, the convergence of biomedical advances with neurotechnology, such as brain–computer interfaces, AI-driven cognitive rehabilitation and personalized neurostimulation, will increasingly connect therapeutic interventions with adaptive learning technologies that respond to individual neurological profiles. In summary, the next 5–10 years will redefine what is possible in maintaining cognitive function, enabling inclusion, and supporting lifelong learning for individuals affected by CNS disorders.
4.2 Innovation supports advancements in Mental-Health and Neurodevelopmental Therapies: Toward Inclu
Neurodevelopmental and mental-health disorders, including attention-deficit/hyperactivity disorder (ADHD), major depressive disorder, anxiety disorders, work-related stress and burnout, impose a substantial and growing global burden. These pressures on individuals, workplaces, and economies underscore the urgent need for more effective, scalable, and accessible interventions.
Improved mental health management can safeguard cognitive capacity, motivation and functional resilience, enabling individuals to pursue education, vocational training and lifelong learning even in the presence of chronic conditions. Scalable and widely accessible digital-health solutions can bridge gaps in service delivery, particularly in low- and middle-income countries where mental-health infrastructure is limited. For institutions such as ITCILO, this evolving landscape presents strategic opportunities to integrate mental-health literacy, digital-health skills and inclusive learning methodologies into capacity-building programs, ensuring that professionals, educators, employers and communities are prepared to leverage innovations responsibly and effectively.
4.3 Smart Drugs for Cognitive Enhancement – Evidence, Uses, and Risks
Beyond physiological concerns, the use of smart drugs raises social and ethical questions. For ITCILO, this trend underscores the need to anticipate ethical guidelines, promote healthfirst learning environments, and develop policies that balance potential benefits with fairness and wellbeing. Preparing adult learners and institutions for this reality will require not only scientific literacy but also strong governance frameworks that ensure enhancements are used responsibly, voluntarily, and inclusively.
4.4 Gamified and citizen-science platforms for learning complex Biotechnology concepts
Gamified environments and citizen-science platforms are emerging as powerful tools to bridge this gap. By transforming complex biotechnology tasks into interactive and engaging challenges, these platforms enable users to explore protein folding, genome annotation, or gene regulation through experiential learning.
By incorporating gamification and citizen-science strategies into adult education, ITCILO can help develop a workforce that is not only more competent in emerging biotechnologies but also more confident and empowered to navigate them as they reshape labour markets and healthcare systems.
4.5 Engaging with personal Biological Data: a new vector for adult learning
We suggest that ITCILO explore and pilot learning models that connect personal data access with guided educational experiences, potentially integrating gamified platforms, citizen-science engagement, and workplace health initiatives. By doing so, the Centre can foster a workforce that is informed, empowered, and prepared to participate in the increasingly data-driven future of health and biotechnology.
Connecting personal genomic data, gamified learning, and inclusion by combining personal data access (e.g., from genetic tests or microbiome profiles) with interactive learning platforms, individuals can move from passive consumers of health information to active learners and informed participants.
4.6 Challenges to address: Data Privacy, Interpretation and Equity
By integrating personal data-enabled learning modules and digital literacy training into its programs, the Centre can strengthen understanding of both biotechnology tools and their implications for privacy, fairness, and ethics. Such initiatives would empower individuals to engage with AI-powered health services responsibly enhancing public trust, supporting equitable implementation, and ensuring that the future of AI in health remains firmly anchored in human dignity and social inclusion.
4.7 Sandbox Experiment Prototype: “MyData BioLab” - Learning with Personal Genomic & Gamified Tools
The proposed sandbox experiment can be interpreted as a response to the growing societal exposure to biological data, which until recently was largely confined to medical institutions and scientific research laboratories. The rationale of the experiment is therefore to prepare constituents to operate in labour markets where biodata becomes more visible, more portable, and more commercially valuable. As part of capacity-building efforts, workers will need to develop an understanding of how their biological data can be generated and used, how informed consent operates in practice, and how to protect themselves from potential misuse or discrimination.
5. Conclusion
For ITCILO, the implications are profound. Emerging biotechnologies will not simply create new technical roles; they will redefine foundational literacies. Adults will increasingly need the ability to interpret probabilistic risk scores, navigate personal biological data, understand the capabilities and limits of AI-generated insights, and recognize the ethical implications of gene editing, data sharing, and synthetic biology. Without inclusive learning ecosystems, these transformations risk deepening existing inequalities, amplifying misinformation, and marginalizing populations with limited digital or scientific access.
At the same time, biotechnology offers powerful opportunities to expand learning and promote social justice. Cloud-based bioinformatics platforms, virtual laboratories, and accessible consumer tools can democratize scientific engagement, while advances in cognitive health, neurodevelopmental therapies, and precision medicine can help restore learning capacities and support lifelong participation for people with disabilities or chronic conditions. The refinement of the ITCILO taxonomy presented here makes these cross-cutting dynamics visible, shifting the focus from siloed application areas to the transversal techniques and data infrastructures that truly shape the future of work.
Looking toward 2030, the central challenge is not technological scarcity but governance, pedagogy, and equitable access. Building capacity in bioethics, data stewardship, interdisciplinary problem-solving, and critical digital literacy will be essential for ensuring that biotechnology strengthens democratic participation, worker agency, and global inclusion.
However, biotechnology innovation remains heavily concentrated in the Global North, where research capacity, investment flows, and regulatory infrastructures are most developed. This geographic concentration poses clear risks for widening global inequalities in access to innovation and its benefits. Limited research, training, and manufacturing capacity in low- and middle-income countries constrains the opportunities identified in this report.
ITCILO is uniquely positioned to lead this transformation, helping Member States build learning systems that are agile, evidence-based, ethically grounded, and capable of empowering all people to engage with the biological dimensions of modern life.
DOWNLOAD FULL REPORT
Report brief by:


